Enzyme inhibition is a fascinating phenomenon that plays a crucial role in understanding biological processes and developing pharmaceutical drugs. Enzymes are proteins that catalyze biochemical reactions in the body, and their activity can be regulated through inhibition. While inhibition may sound negative, it is actually a fundamental mechanism that contributes to the fine-tuning of metabolic pathways and the regulation of cellular functions.
In this article, we will explore 14 captivating facts about enzyme inhibition that will shed light on its importance and applications in various fields. From competitive to non-competitive inhibition, reversible to irreversible inhibition, and allosteric modulation, these facts will unravel the intricate world of enzyme regulation and its impact on drug development, disease treatment, and biochemistry research.
Key Takeaways:
- Enzyme inhibition regulates important body processes and drug development, but can have both therapeutic benefits and adverse effects. It’s a key factor in understanding drug interactions and developing targeted therapies.
- Understanding enzyme inhibition helps scientists develop better drugs and improve our understanding of how our bodies work. Factors like temperature, pH, and other molecules can influence enzyme inhibition.
Enzymes play a crucial role in our body’s biochemical processes.
Enzymes are protein molecules that act as catalysts, speeding up chemical reactions necessary for various physiological functions. They are involved in processes like digestion, metabolism, and DNA replication.
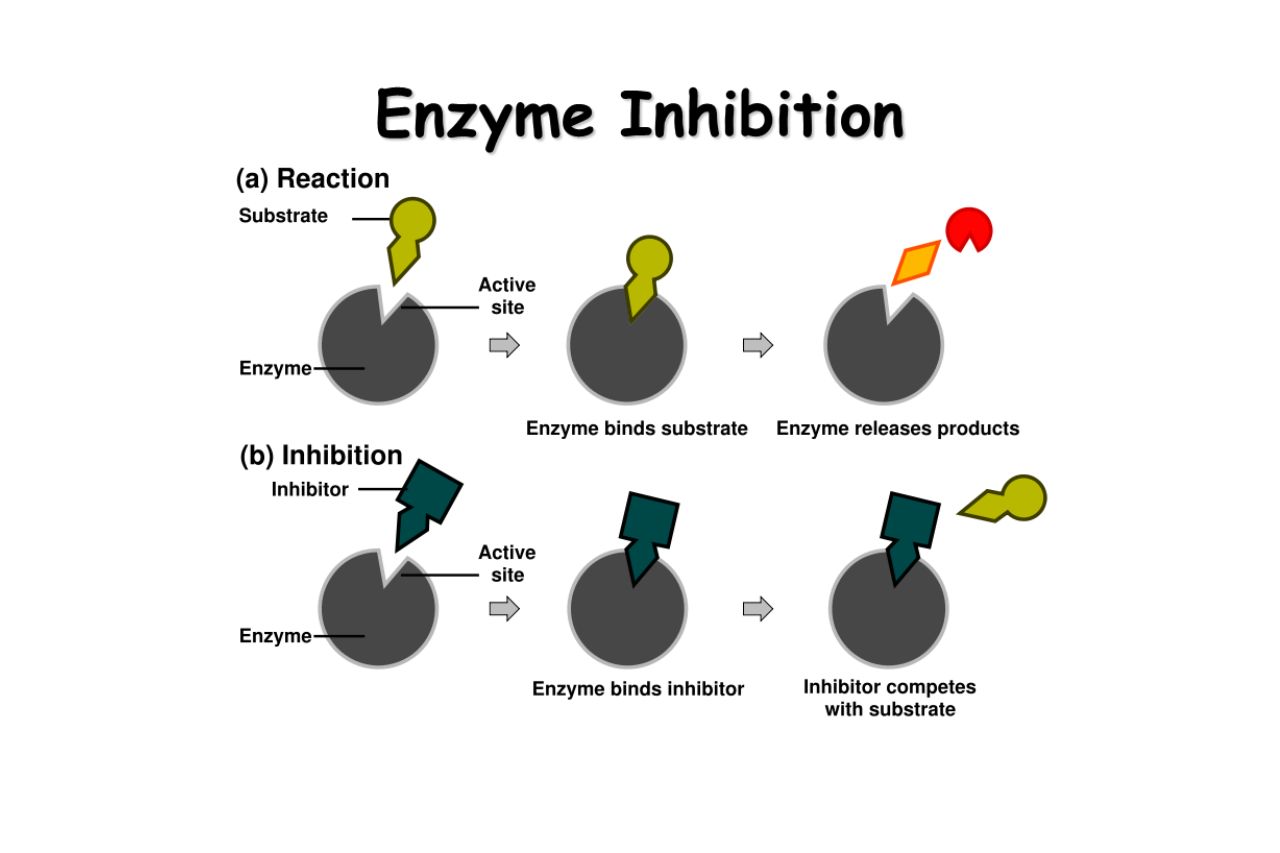
Enzyme inhibition is a regulatory mechanism that controls enzyme activity.
Enzyme inhibition refers to the process of decreasing or stopping the activity of an enzyme. It plays a vital role in maintaining the delicate balance of biochemical reactions in our body.
There are two types of enzyme inhibition: competitive and non-competitive.
In competitive inhibition, a molecule similar in structure to the substrate competes with the substrate for the enzyme’s active site. Non-competitive inhibition, on the other hand, occurs when an inhibitor binds to a different site on the enzyme, altering its shape and preventing substrate binding.
Competitive inhibitors mimic the substrate but do not undergo the chemical reaction.
Competitive inhibitors resemble the substrate in structure, allowing them to bind to the enzyme’s active site. However, they do not undergo the chemical reaction that the substrate would, effectively blocking the enzyme’s function.
Non-competitive inhibitors bind to the enzyme at an allosteric site.
Non-competitive inhibitors bind to an allosteric site, a different site on the enzyme, leading to a conformational change that inhibits the enzyme’s activity. This binding is not affected by substrate concentration.
Enzyme inhibition can be reversible or irreversible.
In reversible inhibition, the inhibitor can bind and unbind from the enzyme. Irreversible inhibition, on the other hand, involves the permanent binding of the inhibitor to the enzyme, rendering it permanently inactive.
Enzyme inhibitors can be natural or synthetic compounds.
Some enzyme inhibitors are naturally occurring substances, such as drugs or toxins, while others are synthetic compounds specifically designed to target certain enzymes.
Enzyme inhibition plays a crucial role in drug development.
Understanding enzyme inhibition is essential in the development of drugs that can target specific enzymes involved in diseases. By inhibiting the activity of these enzymes, the progression of the disease can be slowed or halted.
Enzyme inhibitors can have therapeutic benefits.
Enzyme inhibitors are used in the treatment of various medical conditions, including cancer, high blood pressure, and HIV. By selectively inhibiting specific enzymes, these inhibitors help regulate the processes associated with these diseases.
Enzyme inhibition can have adverse effects.
While enzyme inhibitors can provide therapeutic benefits, they can also have unwanted side effects. Inhibiting enzymes that are involved in essential physiological processes may disrupt normal functioning and lead to adverse effects.
Enzyme inhibitors are extensively studied in medicinal chemistry.
Medicinal chemists extensively study enzyme inhibitors to develop new drugs with improved efficacy and fewer side effects. Understanding the mechanisms of enzyme inhibition helps in the design of better inhibitors.
Enzyme inhibition can be influenced by various factors.
Several factors can influence enzyme inhibition, including temperature, pH, substrate concentration, and the presence of other molecules. Altering these factors can modulate the extent of enzyme inhibition.
Enzyme inhibition is a key factor in understanding drug-drug interactions.
When multiple drugs are simultaneously administered, enzyme inhibition can occur, leading to altered drug metabolism and potential drug interactions. This phenomenon is crucial to consider in clinical settings.
Enzyme inhibition can be reversible with the removal of the inhibitor.
In reversible enzyme inhibition, the inhibitory effect can be reversed by removing the inhibitor or by competitive displacement with a higher concentration of the substrate, allowing the enzyme to regain its activity.
Conclusion
In conclusion, enzyme inhibition is a fascinating phenomenon that plays a significant role in various biological processes. Through the careful regulation of enzyme activity, organisms can control and modulate essential biochemical reactions. From competitive and non-competitive inhibition to reversible and irreversible inhibition, the diverse mechanisms of enzyme inhibition offer a window into the complex world of molecular interactions.Understanding enzyme inhibition is crucial not only in the field of biochemistry but also in drug development and medicine. By studying the effects of inhibitors on specific enzymes, scientists can design drugs that target disease-causing enzymes or enhance the activity of beneficial enzymes.Enzyme inhibition provides valuable insight into the intricate machinery of life at the molecular level. Its study continues to uncover new possibilities for treating diseases, improving industrial processes, and expanding our knowledge of the biological world.
FAQs
1. What is enzyme inhibition?
Enzyme inhibition is a process where the activity of an enzyme is reduced or completely blocked by a molecule known as an inhibitor.
2. What are the different types of enzyme inhibition?
The different types of enzyme inhibition include competitive, non-competitive, uncompetitive, reversible, and irreversible inhibition.
3. How does competitive inhibition work?
In competitive inhibition, the inhibitor molecule competes with the substrate for the active site of the enzyme, thus reducing its activity.
4. What is non-competitive inhibition?
In non-competitive inhibition, the inhibitor binds to a different site on the enzyme, causing a conformational change that reduces the enzyme’s activity.
5. Can enzyme inhibition be reversible?
Yes, enzyme inhibition can be reversible. In reversible inhibition, the inhibitor can bind and unbind from the enzyme.
6. What are irreversible inhibitors?
Irreversible inhibitors bind tightly to the enzyme, forming a covalent bond and permanently blocking its activity.
7. How is enzyme inhibition relevant in medicine?
Understanding enzyme inhibition is crucial in drug development. By targeting specific enzymes, inhibitors can be developed to treat diseases effectively.
8. Are all enzyme inhibitors harmful?
No, not all enzyme inhibitors are harmful. Some inhibitors, such as certain medications, are intentionally used to modulate enzyme activity for therapeutic purposes.
9. Can enzyme inhibition occur naturally?
Yes, enzyme inhibition can occur naturally. Organisms have evolved to produce inhibitors that regulate enzyme activity as part of biological processes.
10. What future developments can be expected in enzyme inhibition research?
Future developments in enzyme inhibition research might focus on discovering novel inhibitors, understanding the intricate mechanisms of inhibition, and applying this knowledge in various fields such as medicine, agriculture, and bioengineering.
Enzyme inhibition is a captivating topic, but there's still more to explore! If you're curious about the intricacies of non-competitive inhibition, our article delving into its enigmatic facts is a must-read. Unraveling the mysteries of enzyme inhibition will leave you with a deeper appreciation for the complexity and importance of these biochemical processes.
Was this page helpful?
Our commitment to delivering trustworthy and engaging content is at the heart of what we do. Each fact on our site is contributed by real users like you, bringing a wealth of diverse insights and information. To ensure the highest standards of accuracy and reliability, our dedicated editors meticulously review each submission. This process guarantees that the facts we share are not only fascinating but also credible. Trust in our commitment to quality and authenticity as you explore and learn with us.


