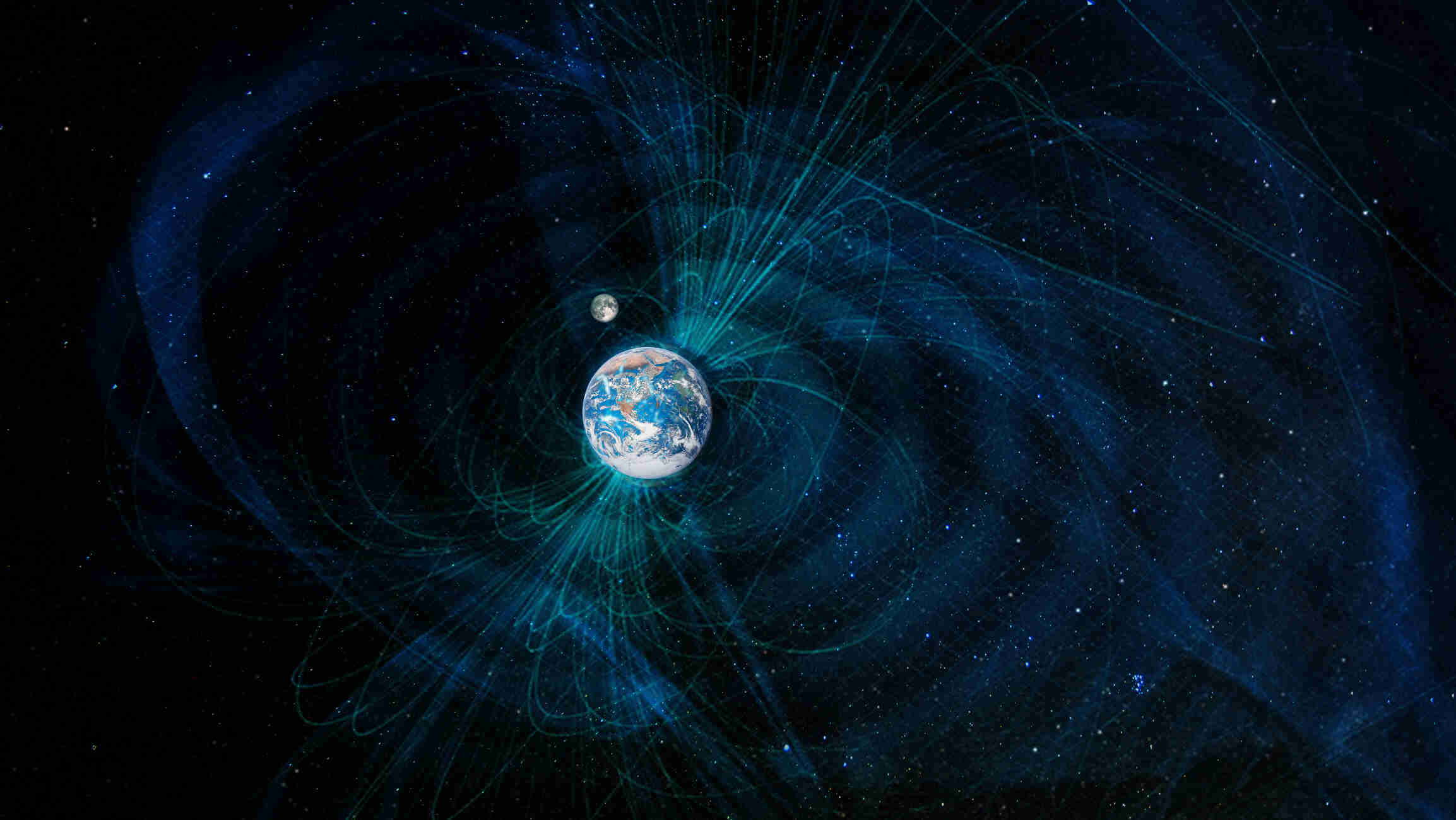
The Earth’s magnetic field is one of the most fascinating and vital features of our planet. It plays a crucial role in protecting life by shielding us from harmful solar radiation and guiding migratory animals to their destinations. But did you know that there is much more to Earth’s magnetic field than meets the eye? From its mysterious origins to its ever-changing nature, the magnetic field holds many intriguing secrets that continue to captivate scientists and researchers around the world. In this article, we will uncover 11 intriguing facts about Earth’s magnetic field that will leave you in awe of this remarkable phenomenon. So, sit back, relax, and let’s dive into the captivating world of the Earth’s magnetic field.
Key Takeaways:
- Earth’s magnetic field is like a protective shield, keeping us safe from solar wind and cosmic radiation. It’s always changing and even creates beautiful auroras in the sky!
- Animals and humans use the Earth’s magnetic field for navigation. Without it, life on Earth would be very different. Scientists study it to understand our planet better.
The Earth has a magnetic field.
The Earth is surrounded by a magnetic field known as the geomagnetic field. It acts like an invisible shield, protecting our planet from solar wind and cosmic radiation.
The magnetic field is not constant.
The Earth’s magnetic field is constantly changing and fluctuating in strength and direction. This phenomenon is known as geomagnetic secular variation.
The magnetic field is generated by the Earth’s core.
The Earth’s magnetic field is generated by the motion of molten iron alloys in the outer core. This motion generates electric currents, which in turn create the magnetic field.
The magnetic field has reversed in the past.
Geological evidence suggests that the Earth’s magnetic field has reversed its polarity multiple times throughout history. These magnetic reversals can be seen in the alignment of magnetic minerals in rocks.
The magnetic field affects navigation.
Many animals, such as birds and sea turtles, can sense the Earth’s magnetic field and use it for navigation during migration. Additionally, humans have used magnetic compasses for centuries to navigate and explore the Earth.
The magnetic field protects us from solar storms.
Solar storms, also known as coronal mass ejections, release highly energized particles that can be harmful to life on Earth. The magnetic field acts as a shield, deflecting and trapping these particles in the magnetosphere.
The magnetic field creates auroras.
When charged particles from the sun interact with the Earth’s magnetic field, they cause beautiful displays of light known as auroras. These stunning natural phenomena can be seen in high-latitude regions such as the northern and southern polar regions.
The strength of the magnetic field varies across the globe.
The strength of the Earth’s magnetic field is not uniform and varies from one location to another. This variation is known as magnetic declination and is an important factor to consider for accurate navigation.
The magnetic field is essential for life on Earth.
The Earth’s magnetic field plays a crucial role in protecting the atmosphere from erosion by solar wind and maintaining a habitable environment for life. Without it, harmful radiation and particles from space would reach the surface.
The magnetic field is used in geophysics.
Scientists and geophysicists use the Earth’s magnetic field to study the interior of the planet. By measuring changes in the magnetic field, they can gain insights into the Earth’s structure and processes.
The magnetic field is constantly monitored.
There are numerous observatories and satellites around the world dedicated to monitoring and studying the Earth’s magnetic field. These efforts help in understanding its behavior and predicting any future changes.
Conclusion
The Earth’s magnetic field is a fascinating and essential aspect of our planet. Its role in protecting us from harmful solar radiation, aiding in navigation, and influencing the behavior of migratory animals is truly remarkable. Understanding the intricacies of this magnetic field can help us gain insights into Earth’s past, present, and future.
From the discovery of magnetic reversals to the mysterious South Atlantic Anomaly, there is still much to learn and explore about Earth’s magnetic field. As we delve deeper into the mysteries of our planet, we continue to appreciate the wonders of nature and the interconnectedness of the Earth’s systems.
So the next time you look at a compass or witness the spectacular dance of the Northern Lights, take a moment to marvel at the incredible phenomenon that is Earth’s magnetic field.
FAQs
1. What causes Earth’s magnetic field?
The Earth’s magnetic field is primarily generated by the movement of molten iron in its outer core.
2. Does Earth’s magnetic field change over time?
Yes, the Earth’s magnetic field changes both in strength and orientation over time. This phenomenon is known as magnetic secular variation.
3. What are magnetic reversals?
Magnetic reversals are when the Earth’s magnetic field flips, causing the magnetic north and south poles to switch places. These events have occurred throughout Earth’s history.
4. How does Earth’s magnetic field protect us from solar radiation?
The Earth’s magnetic field acts as a shield, redirecting and trapping the charged particles from the solar wind and preventing them from reaching the planet’s surface.
5. What is the South Atlantic Anomaly?
The South Atlantic Anomaly is an area where the Earth’s magnetic field is weaker than usual, resulting in an increased risk of damage to satellites and spacecraft passing through it.
6. Can animals sense Earth’s magnetic field?
Yes, many animals, such as birds, sea turtles, and even some insects, have the ability to sense Earth’s magnetic field and use it for navigation during migration.
Was this page helpful?
Our commitment to delivering trustworthy and engaging content is at the heart of what we do. Each fact on our site is contributed by real users like you, bringing a wealth of diverse insights and information. To ensure the highest standards of accuracy and reliability, our dedicated editors meticulously review each submission. This process guarantees that the facts we share are not only fascinating but also credible. Trust in our commitment to quality and authenticity as you explore and learn with us.


