When it comes to the fascinating world of chemistry, there is a wide array of concepts and phenomena to explore. One such concept that holds immense importance in the field is the band gap. The band gap is a fundamental concept in solid-state physics and materials science that has a significant impact on the electrical properties of materials.
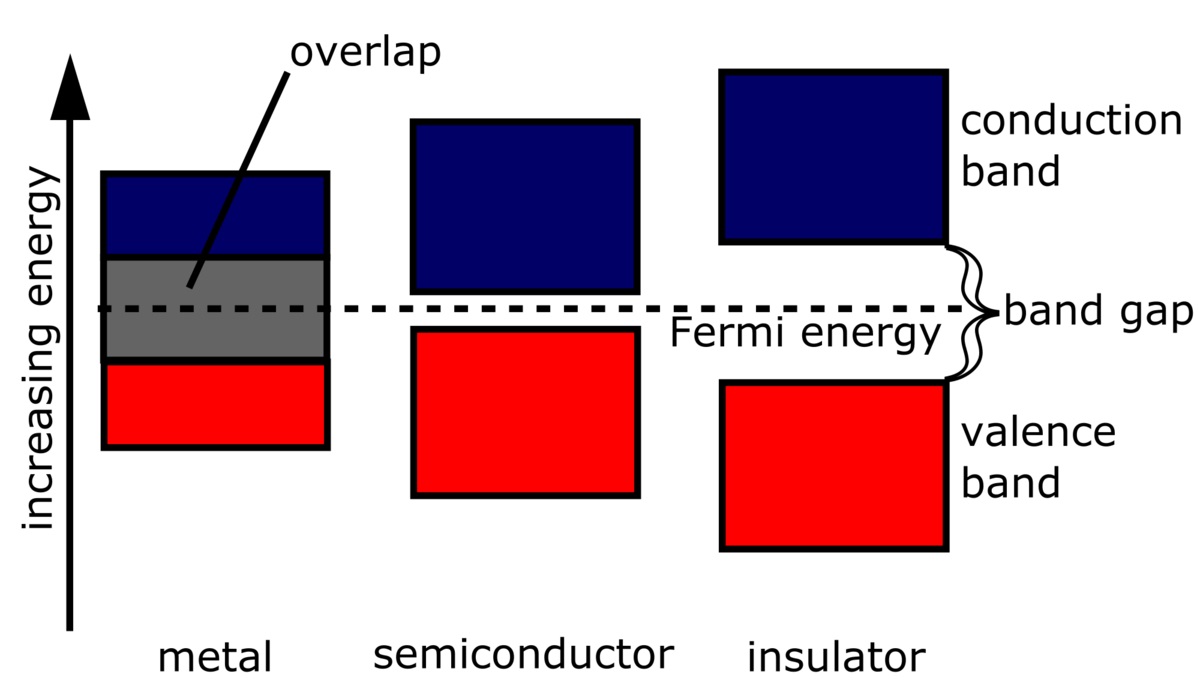
In simple terms, the band gap can be thought of as an energy barrier that exists between the valence band and the conduction band in a material. It determines whether a material is an insulator, a semiconductor, or a conductor.
The study of band gap helps us understand various aspects of materials, such as their conductivity, optical properties, and electronic behavior. In this article, we will delve into the intriguing world of band gaps and uncover some unbelievable facts that will leave you in awe of the remarkable properties of materials.
Key Takeaways:
- Band gap determines if a material conducts, insulates, or acts as a semiconductor. It’s like a material’s superpower that shapes its electrical and optical abilities.
- By tinkering with band gaps, scientists create cool stuff like solar cells and LED lights. It’s like adjusting a material’s talents to make awesome gadgets!
Band gap is a key concept in solid-state physics.
Band gap refers to the energy difference between the valence band and the conduction band of a material. It plays a crucial role in determining the electrical and optical properties of semiconductors and insulators.
The band gap determines whether a material is a conductor, insulator, or semiconductor.
Materials with a small or zero band gap are classified as conductors, while those with a large band gap are insulators. Semiconductors have an intermediate band gap, allowing them to conduct electricity under certain conditions.
Band gap engineering is essential for optimizing electronic devices.
By manipulating the band gap of a material through various techniques such as alloying, doping, and quantum confinement, researchers can tailor the properties of semiconductors for specific applications like photovoltaics, LEDs, and transistors.
The band gap of a material affects its absorption and emission of light.
Materials with a larger band gap absorb and emit higher-energy photons, making them suitable for applications such as ultraviolet light detection and high-efficiency solar cells.
The band gap of a material can be measured experimentally using various techniques.
Methods like optical absorption spectroscopy, photoemission spectroscopy, and electrical conductivity measurements can provide valuable insights into the band structure and band gap of a material.
Band gap decreases with increasing temperature in most materials.
As the temperature rises, thermal energy promotes electrons from the valence band to the conduction band, reducing the effective band gap and increasing the material’s conductivity.
The band gap of graphene is zero, making it an exceptional conductor.
Graphene, a single layer of carbon atoms arranged in a hexagonal lattice, possesses a unique electronic structure with no band gap. This property gives it remarkable conductivity and makes it a promising material for future electronic devices.
Quantum dots are nanoscale structures with tunable band gaps.
Quantum dots are tiny particles that exhibit quantum confinement effects, leading to discrete energy levels and a size-dependent band gap. Their unique properties make them valuable for applications in optoelectronics and quantum computing.
Band gap engineering can be achieved through strain manipulation.
By subjecting materials to mechanical strain, either compressive or tensile, it is possible to modulate their band gap. This technique has found applications in flexible electronics and strain sensors.
Band gap variations can result in different colors of LED lights.
The energy band gap directly influences the wavelength of light emitted by LEDs. By selecting specific materials with different band gaps, LEDs can produce a wide range of colors.
The concept of indirect and direct band gaps is crucial in semiconductor physics.
In materials with indirect band gaps, electrons need an additional energy boost to transition between the valence and conduction bands. In contrast, direct band gap materials allow for efficient electron transitions, making them more suitable for optoelectronic applications.
Band gap narrowing is a phenomenon observed in heavily doped semiconductors.
When a semiconductor is heavily doped with impurities, the increased concentration of charge carriers can cause the effective band gap to shrink. This effect is known as band gap narrowing.
Band gap plays a crucial role in the design of solar cells.
Solar cells rely on photon absorption to generate electricity. The band gap of the semiconductor material used in solar cells determines the range of wavelengths of light that can be efficiently converted into electrical energy.
Band gap engineering is utilized in the development of advanced optoelectronic devices.
By precisely controlling the band gap of materials, researchers can create optoelectronic devices with enhanced efficiency, such as high-speed photodetectors, laser diodes, and OLED displays.
Band gap measurements are crucial for quality control in semiconductor manufacturing.
Accurate determination of the band gap ensures the consistency and reliability of semiconductor devices produced in large quantities, such as integrated circuits and microprocessors.
The band gap of a material can be influenced by external factors, such as electric fields.
By applying an electric field, it is possible to modulate the band structure of certain materials, resulting in a change in their band gap. This effect is utilized in devices like field-effect transistors (FETs).
Band gaps are not only limited to inorganic materials but also found in organic semiconductors.
Organic semiconductors are compounds composed of carbon and hydrogen atoms, and they exhibit unique electronic properties, including tunable band gaps. This makes them suitable for applications in organic electronics, such as flexible displays and organic solar cells.
The understanding and manipulation of band gaps are essential for advancing modern technology.
From the development of more efficient solar cells to the creation of faster and smaller electronic devices, the ability to engineer and control band gaps opens up new horizons in the field of materials science and technology.
Conclusion
In conclusion, the band gap is a fascinating and complex concept in the field of chemistry. It plays a crucial role in determining the electrical and optical properties of materials. Understanding the band gap is essential for various applications, including the development of semiconductors, solar cells, and electronic devices.Throughout this article, we have explored 18 unbelievable facts about the band gap. From the discovery of the band gap to its manipulation through doping and the impact on material conductivity, we have delved into the intricacies and significance of this phenomenon.From a better understanding of band gaps, scientists and researchers can continue to push the boundaries of material science and technological advancements. As we uncover more about the band gap mechanism, we can harness its potential for creating innovative and energy-efficient solutions.So, the next time you encounter the term “band gap,” remember its importance in shaping the properties of materials and driving technological innovation. The band gap truly is a fascinating concept that continues to unlock new possibilities in the world of chemistry and beyond.
FAQs
Q: What is a band gap?
A: The band gap is the energy difference between the valence band and the conduction band in a material. It represents the minimum energy required for an electron to move from the valence band (occupied by electrons) to the conduction band (unoccupied by electrons).Q: Why is the band gap important?
A: The band gap is crucial because it determines the electrical and optical properties of a material. It influences the conductivity, semiconductor behavior, and the ability to absorb or emit light.Q: How is the band gap determined?
A: The band gap is determined through experimental techniques such as spectroscopy, which measures the absorption or emission of light by a material. It can also be calculated using theoretical models and simulations.Q: Can the band gap be modified?
A: Yes, the band gap can be modified through a process called doping. By introducing impurities into a material, scientists can alter the band structure and manipulate the band gap to achieve desired properties.Q: What are the applications of the band gap?
A: The band gap has numerous applications, including the development of semiconductors for electronic devices, solar cells for harnessing solar energy, and optoelectronic devices such as LEDs and lasers.Q: Are all materials with a band gap semiconductors?
A: No, not all materials with a band gap are semiconductors. Depending on the size of the band gap, materials can be classified as insulators (large band gap), semiconductors (moderate band gap), or conductors (no band gap).Q: Are there any materials with zero band gap?
A: Yes, there are materials known as zero-gap or zero-band materials that have a band gap close to zero. These materials exhibit unique properties and are of great interest in the field of physics and materials science.
Band gaps are truly fascinating, but the rabbit hole goes even deeper. Want to learn more about the intricacies of semiconductor physics? Explore the mind-blowing facts about indirect band gap semiconductors and unravel the mysteries behind their unique properties. From the fundamentals to cutting-edge applications, there's always more to discover in the captivating world of band gaps.
Was this page helpful?
Our commitment to delivering trustworthy and engaging content is at the heart of what we do. Each fact on our site is contributed by real users like you, bringing a wealth of diverse insights and information. To ensure the highest standards of accuracy and reliability, our dedicated editors meticulously review each submission. This process guarantees that the facts we share are not only fascinating but also credible. Trust in our commitment to quality and authenticity as you explore and learn with us.


